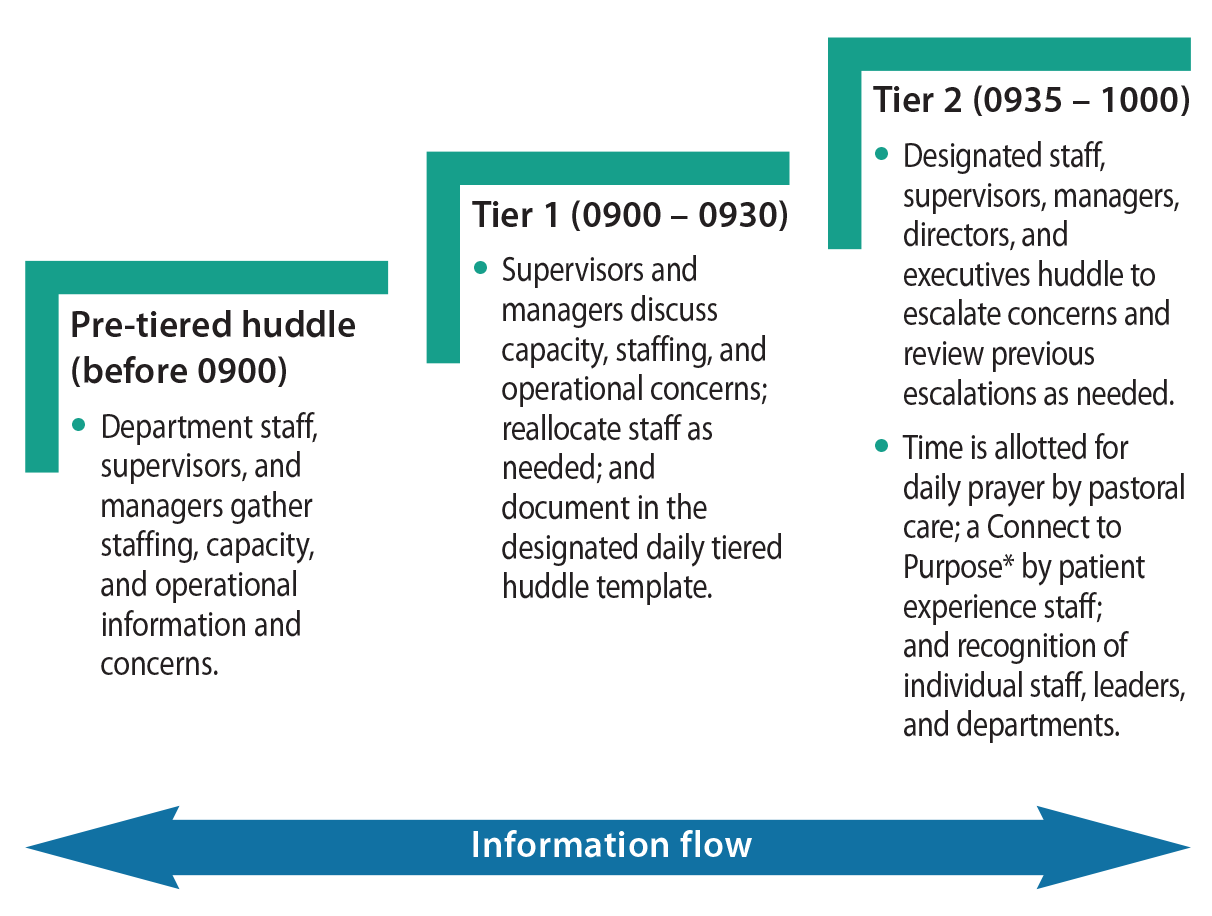
Consortium's next conference on technology and nursing to take place at Disney's Coronado Springs ResortEarly-bird registration now open Sponsorships opportunities available The Nursing Consortium of Florida's next conference Caring Connections; Discovering Magic at the Intersection of Technology & Nursing will take place at Disney’s Coronado Springs Resort. The two half-day conference will begin at noon on Friday June 28 and conclude before noon the following day. Register today and be among the first to see the special travel packages available only to conference attendees and their accompanying families and friends. Become a conference sponsor and add to the magic! In addition to a great conference, we will offer an extraordinary get-a-way value for those traveling to Orlando and locals alike. Once registering to attend the conference, attendees will be able to book a discounted hotel room and select from a range of specially-priced Theme Park tickets including partial-day tickets available only to those attending a conference at Walt Disney World®. Kaufman Hall reports on improving hospital Average Length of StayThe timely discharge of patients to skilled nursing facilities (SNFs) remains a significant concern for health system leaders. There have been some signs of improvement: just over 40% of respondents to Kaufman Hall’s 2023 State of Healthcare Performance Improvement survey indicated that inpatient average length of stay (ALOS) had decreased over the past year. But that follows the results of Kaufman Hall’s 2022 survey, in which nearly 70% of respondents reported an increase in inpatient ALOS. For a majority of the 2023 respondents, inpatient ALOS had either not improved or had gotten worse over the past year. Kaufmann Hall says ensuring that inpatient discharge processes are optimized will remain an important focus of health system performance improvement efforts in reducing ALOS. But these efforts will be diminished if there is not a sufficient supply of SNF beds available to receive patients discharged from inpatient care. To successfully bring down ALOS, health system management will need to combine performance improvement efforts with efforts to successfully align themselves more closely with skilled nursing providers. Please click here to read more. Cleveland Clinic and IBM researchers publish findings on Artificial Intelligence and ImmunityResearchers from Cleveland Clinic and IBM have published a strategy in Briefings in Bioinformatics for identifying new targets for immunotherapy through artificial intelligence (AI). This is the first peer-reviewed publication from the two organizations’ partnership, Discovery Accelerator, designed to advance research in healthcare and life sciences. Timothy Chan, M.D., Ph.D., chair of Cleveland Clinic’s Center for Immunotherapy and Precision Immuno-Oncology, says “Partnering with IBM allows us to push the boundaries of artificial intelligence and health sciences research to change the way we develop and evaluate targets for cancer therapy.” For decades, scientists have been researching how to better identify antigens and use them to attack cancer cells or cells infected with viruses. This task has proved challenging because antigen peptides interact with immune cells based on specific features on the surface of the cells, a process which is still not well understood. Research has been limited by the sheer number of variables that affect how immune systems recognize these targets. Identifying these variables is difficult and time intensive with regular computing, so current models are limited and at times inaccurate. Please click here to read more Miami Cancer Institute now offers some cancer patients robotic surgery through a single incision.jpg) Through a single incision of approximately one inch, doctors at Consumer member Baptist Health Miami Cancer Institute are now able to perform robot-assisted surgery on some patients with head and neck cancers, urologic cancers, and soon, gynecologic cancers. The single-port technology is part of Baptist Health’s robotic surgery program, one of the highest-volume programs in the country. Minimally invasive surgery through the robotic approach has really revolutionized how we perform surgery, and with this novel technology we now have better access to more narrowed areas of the body,” said John Diaz, M.D., chief of gynecologic oncology at the Institute. Dr. Diaz, who is also director of robotic surgery and chair of the Robotics and Innovations in Surgery Subcommittee at Baptist Health, said that as the evolution of robotics continues, the number of patients who can benefit from the technology will expand even further. Please click here to read more. How tiered huddles help improve operations at Miami Cancer InstituteConsortium member Miami Cancer Institute’s (MCI) Tiered Huddles program was recently featured in a front-page article of NURSLINE, published by the American Nurse Journal. The story shows how MCI has successfully implemented “tiered huddles” -- brief conversations that occur throughout levels of an organization or across health systems to improve patient safety, communication, situational awareness, care coordination, resource utilization, patient outcomes, and service.
Using an inclusive approach, MCI constructed a meaningful process with measurable operational and patient-centered metrics. In keeping with leadership feedback regarding tiered huddle design, we created a simple Excel report template with drop-down fields to reduce documentation variation and simplify report generation and analysis. Each department leader or their staff designee completed one section, labeled with their name, department, and floor location. We placed the monthly report template and agenda in Microsoft Teams, to which all clinical and operational leaders, their staff designees, and executives had access. Please click here to read more. Nation’s largest associations of nurses team up to offer new course on ethicsThe National League for Nursing and the American Nurses Association, two of the nation’s leading nursing professional associations, have joined forces to offer Ethics in Nursing Education: Tools for Cultivating Ethical Competence in Learners. This is a new, groundbreaking initiative for nurse educators across the spectrum of higher education that focuses on how to best incorporate ethics into the nursing curriculum. As nurses today encounter an increasingly diverse population of patients under their care and a diversity of backgrounds among their health care colleagues, awareness of multiple perspectives and cultivating a moral sensibility is more vital than ever. In any health care setting, a nurse’s decision to provide culturally competent care and health care services—or a refusal or failure to provide care—will undoubtedly have consequences; some anticipated, some not. Understanding the implications of those decisions is crucial to advance a culturally sensitive, ethical approach to caregiving to maximize health outcomes. The discipline of ethics, as presented here in eight sessions, uses cognitive research developer James Rest’s respected multi-step model for ethical decision-making to explore implications for scholarly research, administration, and nursing practice. Please click here to read more. |
McKinsey Health Institute reports on the case for closing the women’s health gapA new report by the McKinsey Health Institute and the World Economic Forum on the gender health gap found this gap equates to 75 million years of life lost due to poor health or early death. Addressing it could boost the global economy by at least $1 trillion annually the report says. While women live longer on average, they face a variety of health disadvantages compared with men and spend 25 percent more of their lives in poor health. Fixing this could add seven healthy days per woman per year on average, leading to longer lives and more days spent in good health. The report says women with very serious health conditions wind up lost in health systems for years. The analysis is based on a detailed assessment of the 64 conditions that account for more than 85 percent of the female global disease burden in total. In analyzing more than 650 academic research papers, the report found that close to half didn’t provide data on sex-based differences. “This data gap leads to a lack of understanding of how medicine and science work for women, and it contributes to their often receiving substandard care,” says McKinsey partner and MHI report co-author Anouk Petersen. Please click here to read more. Evening of Gratitude dinner raises $400,000 for Bethesda Hospital Emergency ServicesOver 150 guests attended the Evening of Gratitude dinner benefiting Consortium member Bethesda Hospital in Boynton Beach, which was held in January 2024 at the OPAL Grand Resort & Spa in Delray Beach. Hosted by Baptist Health Foundation, the event honored members of its Giving Society and other donors to Bethesda Hospital. The funds will used to renovate the Emergency Department at Bethesda Hospital East. “The Emergency Department is often the front door of the hospital for many in our community. Patients and their families depend on the expert care of our emergency medicine physicians, nurses and staff, as well as our equipment and technology,” said Barbara James, Baptist Health Foundation vice president…As a not-for-profit organization, we rely on the generosity of donors to Baptist Health Foundation to help support world-class, compassionate care, close to home.” Please click here to read more. FAU earns ANCC accreditation for Nursing Continuing Professional DevelopmentConsortium member Florida Atlantic University’s Christine E. Lynn College of Nursing has achieved accreditation from the American Nurses Credentialing Center (ANCC) as a provider of Nursing Continuing Professional Development (NCPD). This prestigious recognition demonstrates the college’s dedication to providing the highest quality of professional development for registered nurses and the highest standards of excellence in the delivery of health care. “The American Nurses Credentialing Center accreditation process represents the gold standard for continuing education for nurses and we are incredibly proud to receive this significant recognition,” said Louise Aurélien-Buie, Ed.D., assistant dean of executive and continuing education and academic-practice partnerships, FAU Christine E. Lynn College of Nursing. “Achieving accreditation is a testament to our continued commitment to advancing compassionate patient care through clinical practice, education, research and innovation.” Please click here to read more. Holy Cross Health receives $2 million gift from Jon and Helaine Ayers for Outpatient Rehabilitation CenterConsortium member Holy Cross Health has announced that Fort Lauderdale residents Jon and Helaine Ayers have made a $2 million gift to support the hospital’s Outpatient Rehabilitation Center. In recognition of the generous gift, the center will be renamed the Jon and Helaine Ayers Rehabilitation Institute. With this gift, the Rehabilitation Institute will undergo a transformational physical renovation and the addition of significant investments in the latest state-of-the-art rehabilitation equipment. ”We are blessed to have the support of Jon and Helaine Ayers, who are committed to the highest quality compassionate care for all in our community,” said Mark Doyle, President and CEO of Holy Cross Health. Rehabilitation hits close to home for Jon, the former Chairman and CEO of IDEXX Laboratories, a global leader in pet healthcare innovation. In June 2019, Jon was seriously injured in a catastrophic bike crash, caused by another cyclist, leaving him a quadriplegic. “My accident reshaped my outlook on life. I continue to spend roughly 20 hours a week in physical therapy, working to improve strength, function and wellbeing,” Jon said. “I am so appreciative of the care I have received, and I want to make sure other patients have access to great therapists, supported by the latest technology, treatments and equipment.” Please click here to read more. Anti-trafficking experts advocate victim-centered response at UM SONHS event on Human Trafficking“We Can All Do Something” was the theme of a recent educational event about dealing with human trafficking in healthcare, presented recently by Consortium member University of Miami’s School of Nursing and Health Studies. A distinguished panel of anti-trafficking advocates shared their expertise during the program, “Human Trafficking Response — A Conversation.” The panelists, who hail from diverse law enforcement and health care backgrounds, discussed their own stories of commitment to human trafficking awareness education. They emphasized the importance of taking a victim-centered, trauma-informed approach as well as accepting personal responsibility for changing the culture that has allowed human trafficking to thrive in our midst. Dean Cindy L. Munro said, “Health care providers, no matter where they work or live, are very likely to come in contact with human trafficking victims. It is imperative that nurses and all health care providers are able to recognize people impacted by human trafficking and respond appropriately to their extensive, complex, often urgent needs.” One attendee, a health science major, said “I learned a lot more facts, and I feel more educated about the topic. I’m happy I now know the phone number [human trafficking hotline, 888-3737-888] in case I see something.” Please click here to read more. Nicklaus Children's Hospital launches Safe+Sound initiative to protect kids from digital technologyTo support parents in navigating the complexities of today's digital age, in which children can access inappropriate content, experience cyberbullying, suffer mental distress and addiction by using their screen devices, Consortium member Nicklaus Children's Hospital, in partnership with the Digital Health Institute for Transformation (DHIT) and other key community partners has announced the formation of The Digital Safety Alliance and launched the Safe+Sound initiative which provides information and resources regarding the impact and long-term effects of digital technology – including smartphones, tablets, computers and gaming devices – on the well-being of children and families, as research shows a growing correlation between internet and social media access by children and an increase in suicidal thoughts, depression, psychological distress, eating disorders, disrupted sleep patterns, addiction and other health-related concerns. The Safe+Sound awareness campaign includes a website with vetted information, news articles, blogs, and research that parents, guardians, educators, and healthcare professionals can access with extensive data and helpful information. Please click here to read more. HHS alerts health sector to 21 new cyber vulnerabilities
Nicklaus Children's Hospital receives historic gift from Citadel founder and CEO Kenneth C. GriffinConsortium member Nicklaus Children’s Hospital has received one of the largest single donations in the hospital’s 72-year history. The $25 million gift from Citadel founder and CEO Kenneth C. Griffin will support the new five-story, 127,000-square-foot surgical tower opening in 2024. In recognition of this historic gift, the tower will be named the Kenneth C. Griffin Surgical Tower. Mr. Griffin’s donation will also help fund Nicklaus Children’s four major institutes advancing pediatric care related to the brain, cancer and blood disorders, the heart, and orthopedics. “This transformational gift will change the future of pediatric care for generations to come and ensure that doctors, nurses, and specialists have the latest technological advances at their fingertips to treat the children we serve,” said Matthew A. Love, president and CEO of Nicklaus Children’s Health System, the hospital’s parent organization.“ Please click here to read more. |


![cssheader=[tonheaderclass] cssbody=[tonbodyclass] header=[More about]body=[] More about](modules/News/css/images/transparent.gif)

 The health care sector should quickly implement patches or mitigations to address 21 new cyber vulnerabilities identified by the Cybersecurity and Infrastructure Security Agency in January, the Department of Health and Human Services’ Health Sector Cybersecurity Coordination Center (HC3) recently
The health care sector should quickly implement patches or mitigations to address 21 new cyber vulnerabilities identified by the Cybersecurity and Infrastructure Security Agency in January, the Department of Health and Human Services’ Health Sector Cybersecurity Coordination Center (HC3) recently 